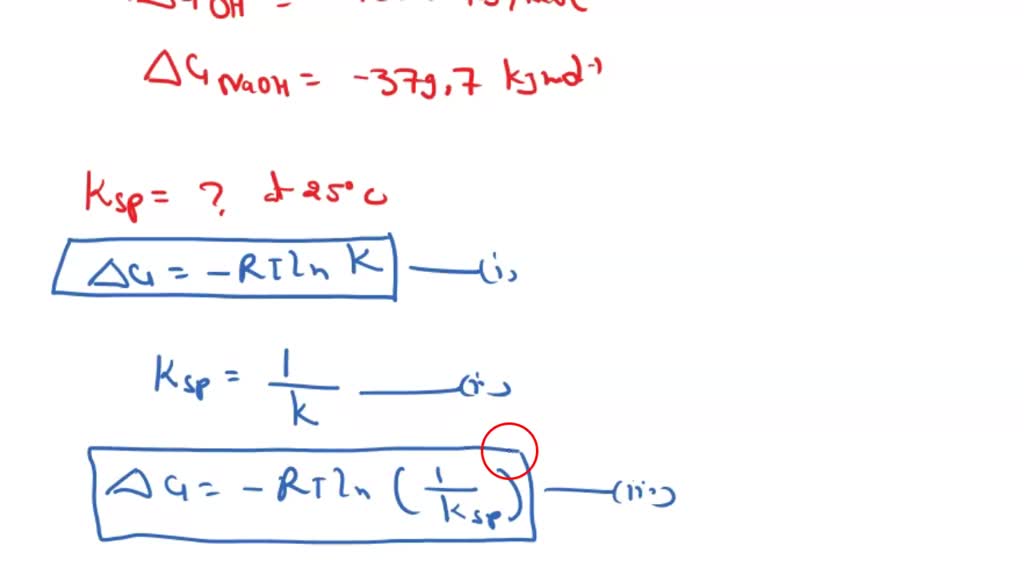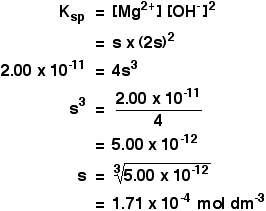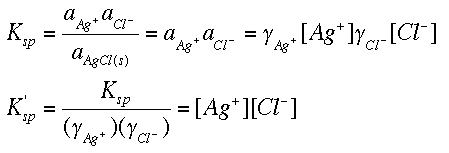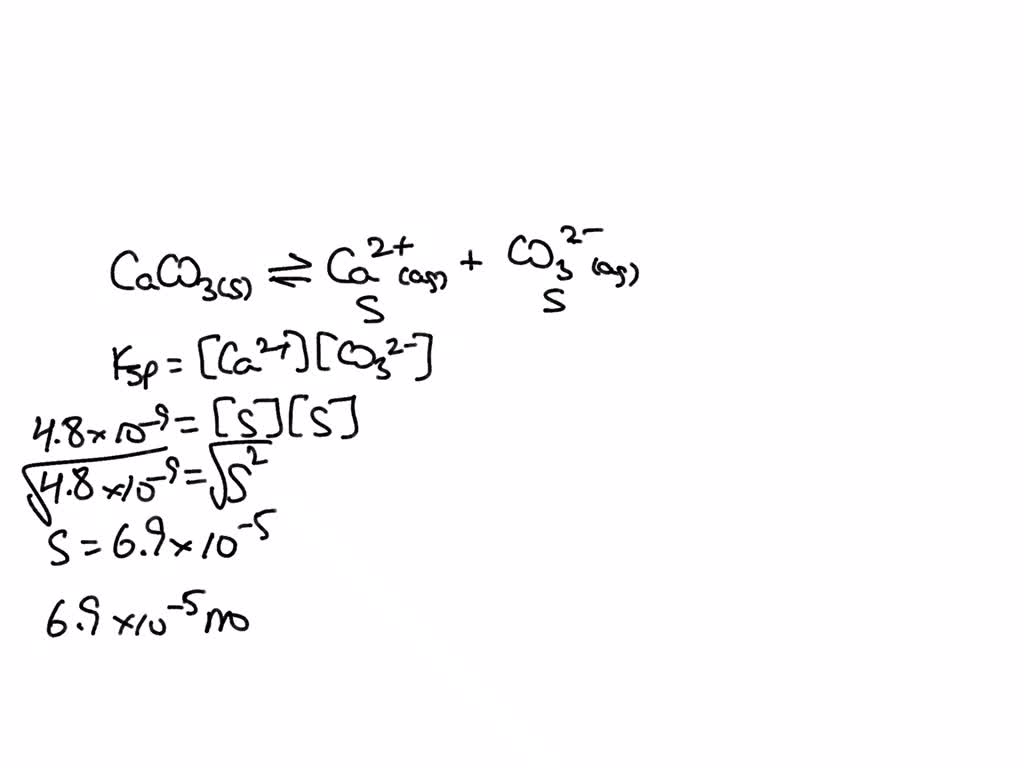
SOLVED: Calculate the solubility of calcium carbonate (CaCO3) in g/L. (Ksp, CaCO3 = 4.8x10^-9) (Mm, CaCO3 = 100.9 g/mol) A) 4.8x10^-10 B) 6.9x10^-5 C) 4.8x10^-9 D) 6.9x10^-3

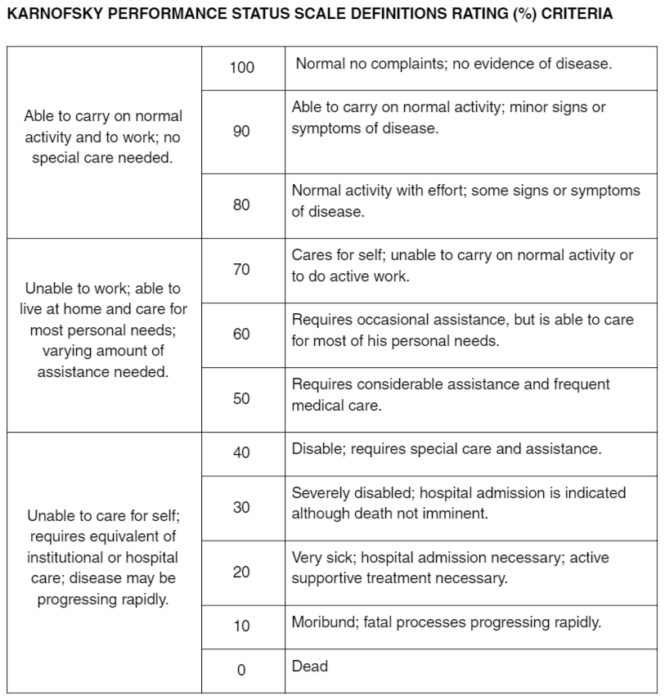
Christopher Steer on Twitter: "Hui- Online calculator https://t.co/X7sESMwiCE Is it the https://t.co/xOY5njAIC8 for #pallonc? #MASCC18 #prognostication workshop #supponc #gerionc https://t.co/XCET9YeVh5" / Twitter

SOLVED: The solubility of calcium sulfate (CaSO4) is 0.67 g/L. Calculate the value of Kps for calcium sulfate. If we know the solubility of CaSO4 and are asked to calculate his Kps.